by Sérgio Fontinhas. What’s the best ergogenic aid? Is it creatine? Caffeine? Vitamin D?
If you answered ‘water,’ then you’re clearly a Mensa member.
The human body is approximately 60% to 70% water (with a range of 45-75%) (1). It can be is less with increasing body fat because fat is known as “anhydrous” with about 10% water, however fat-free mass can be 70-80% water (1). An average 70-kg person has approximately 42 L of total body water, with a range of 31–51 L (1). Improper hydration will result in either dehydration or overhydration (hyponatremia). Daily water balance depends on the net difference between water gain and water loss.
Individuals are routinely at a risk of mild dehydration day to day (2). Public surveys (10,11) and experimental trials (12,13) indicate that the general public, and most importantly special populations such as children and older adults, are at a risk of voluntary dehydration (14,15). Even experienced athletes can underestimate their hydration status and may drink insufficient amounts of water resulting in sustained dehydration (16).
Sustained dehydration is associated with poor health (3,4) and increases the likelihood of kidney stones and urinary tract infection by a significant degree (3,5). Additionally, prolonged vasoconstriction due to chronic dehydration increase the risk of hypertension and stroke (6).
An emergent body of evidence also suggests water consumption (and the food we eat) affect mental and physical performance (7). Water is essential to the maintenance of normal physical and cognitive function (8), and there are some recommended intake guidelines of 2000 ml of fluids for females and 2500 ml for males per day (9). More specifically it is recommended 3.7 L for 70 kg males and 2.7 L for 57 kg females (29).
Water is the medium of circulatory function, biochemical reactions, metabolism, substrate transport across cellular membranes, temperature regulation, and numerous other physiological processes (28). The loss or increase in fluids and electrolytes (potassium, sodium, calcium, magnesium…) affects cellular performance, and can cause cell death, and even death of the entire organism.
Assessing hydration status – Individuals should monitor their own hydration levels using markers such as urine color (27). Although dilution methods to determine total body water via plasma osmolality measurement are the most accurate, valid, and sensitive ways, they are not practical for most situations (30,122), but total body mass and urine color when used in conjunction is a good way to assess hydration status (30,122).
First-morning urine should look like pale yellow (27), indicating the normal and expected presence of some waste products from metabolism overnight. This color corresponds to a state of euhydration (34,35). I shouldn´t look at water, or anything dark.
Thirst is initially perceived when a body weight deficit of 1–2% exists (36,37), fluid consumption should be adequate to avert the perception of thirst. Thirst signals any imbalances of the osmolality of fluids and tissues (the electrolyte concentration), and the total amount of water in our body (volume).
Dehydration – Dehydration is characterized by weight loss, confusion, dry skin that is hot to the touch, and possibly an elevated core body temperature. In a hot climate, dehydration can be dangerous and result in thermal injury. Other causes of dehydration can be excess diarrhea, vomiting due to GI dysfunction, kidney disease, and diuretic medications.
Thirst may be a poor measure of hydration because of the lag between the physiological dehydration and the thirst signal. Special populations require more attention, elderly are less sensitive to the thirst mechanism the deterioration of osmoreceptor sensitivity in older adults (30,38,39,40,121); and children are inexperienced in interpreting the thirst response (41,42). Older adults are also at higher risk for reduced kidney filtration function, which results in less efficient water conservation (when dehydrated), further exacerbating difficulties in recognizing a dehydrated state (43).
Physiology of dehydration / physiological response
When the body is in a state of dehydration, many substrates and neurotransmitters are influenced by circulating vasopressin (antidiuretic hormone) and angiotensin II (44,45). Dehydration can increase levels of cortisol (46). Interestingly, even a decrease in cell volume caused by hypohydration promotes insulin resistance (47,48,49).
Conditions dehydrating insulin target tissues such as hyperosmolarity or amino acid deprivation are associated with insulin resistance; blockage of the cell volume response to insulin may be the common denominator in dehydration-induced insulin resistance (47).
As a consequence of dehydration, the blood–brain barrier permeability is altered by serotonergic and dopaminergic systems, potentially causing central nervous system dysfunction if dehydration is prolonged (50). Chronic dehydration influence inhibitory and excitatory activities of the brain by increasing aminobutyric acid and glutamate levels (51), by stimulating γ-aminobutyric acid and N-methyl-D-aspartate receptors, to synthesize and release antidiuretic hormone (56).
Even mild dehydration produces significant changes at the neural level: total brain volume shrinkage and over-recruitment of specific brain areas during cognitively demanding tasks (52).
Hypohydration and exercise
Hypohydration during exercise strongly rises the catabolic hormonal response to resistance exercise, and increase circulating concentrations of metabolic substrates. Hydration status during exercise changes the endocrine and metabolic adaptive responses to resistance exercise and also changes the postexercise internal environment. Specifically, there’s increase in cortisol, epinephrine, and norepinephrine (46).
Hypohydration stimulates the catabolic hormones by increasing core temperature (69,70) and increases cardiovascular demand due to decreased plasma volume (71,72,73). A 3% to 4% loss of body weight (water) reduces strength by about 2% and power by about 3% (35).
As noted before, a decrease in cell volume caused by hypohydration promotes insulin resistance (47,48,49). Resistance exercise might exacerbate the effects of hypohydration on insulin resistance, because muscle damage is also related with insulin resistance (74,75,76,77). There’s decreased GLUT-4 protein content (78,79), and impaired insulin signal transduction at the level of IRS-1, PI 3-kinase, and Akt-kinase (75). Downhill treadmill running and resistance exercise result in transient insulin resistance (80,81,82), and the reductions in glucose uptake in muscle damage models may be of the order of 20–30% (78,80).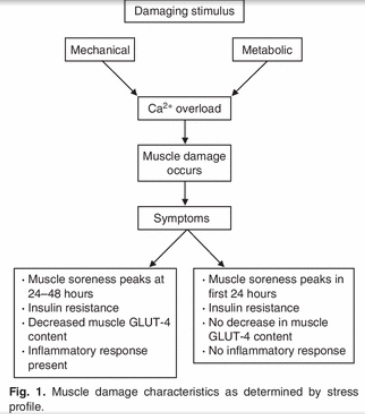
(74).
Hyponatremia – Extreme exercise conditions (equal or above three hours continuously), such as the marathon or triathlon, without the intake of electrolytes increase the risk dehydration or hyponatremia (83). Symptomatic hyponatremia is typically observed with greater than 6 hours of prolonged exercise. Acute water toxicity happens due to rapid consumption of large quantities of water that greatly exceeded the kidney’s maximal excretion rate (from 0.7 to 1.0 L/hour) (1).
Severe exercise-associated hyponatremia (EAH) starts as significant mental status changes resulting from cerebral edema, at times associated with noncardiogenic pulmonary edema (84,85). The osmotic imbalance results in fluid movement into the brain, causes swelling, which then leads to disorientation, confusion, general weakness, grand mal seizures, coma, and possibly death (35,124,125,126).
Exercise-associated hyponatremia (EAH) typically occurs during or up to 24 hours after prolonged physical activity, and is defined by a serum or plasma sodium concentration below the normal reference range of 135 mEq/L (86,87).
Usually only less than 1% of marathons athletes present signs of EAH (88,89), however it was as high as 23% in an Ironman Triathlon (90) and 38% in runners participating in a marathon and ultramarathon in Asia (91). There’s also now a trend for symptomatic EAH for shorter distance events, such as a half marathon (92) and sprint triathlon taking approximately 90 minutes to complete (93). There’s also no statistical significance for the incidence of symptomatic between genders (55), though women may be at greater risk than men (55).
We have seen that the major risk factor for developing EAH is excessive water intake beyond the capacity for renal water excretion (1,100,101) largely as a result of persistent secretion of arginine vasopressin (102,103). Elevations in brain natriuretic peptide (NT-BNP) may lead to excessive losses of urine sodium and raise the risk of hyponatremia (104).
Another concern is the inability to mobilize body sodium bound in bone. Sodium can be released from internal stores such as bone (105,106,107), 25% of body sodium is bound in bone (osmotically inactive) and is potentially recruitable. Inability to recruit sodium from that pool may increase the risk of hyponatremia.
Individuals under normal conditions are able to excrete between 500 and 1000 mL/h of water (108), plus the non-renal losses of water as sweat, so athletes should be able to consume as much as 1000 to 1500 ml/h before developing water retention and dilutional hyponatremia, therefore it seems likely that excessive water intake (>1500 mL/h) is the main cause.
The combination of excessive water intake and inappropriate AVP secretion will clearly lead to hyponatremia.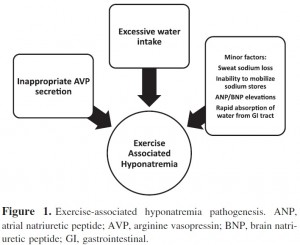
(86)
Arginine vasopressin (AVP) must be suppressed appropriately with water loading, otherwise the ability to produce dilute urine is markedly impaired (109).
Water can also be absorbed from the gastrointestinal tract at the end of a race causing an acute drop in serum sodium concentration (110), with clinical signs of EAHE after 30 minutes. During exercise, breakdown of glycogen into lactate increases cellular osmolality and rises serum sodium, but some minutes after exercise this is reversed and serum sodium levels drop (111,112).
The risk also rises if the degree of fluid loss through sweat is sufficient to produce significant volume depletion (stimulating AVP release and impairing urine excretion of water), coupled with ingestion of hypotonic fluids (86).
Although not conclusive, nonsteroidal anti-inflammatory drugs (NSAIDs) have been implicated as a risk factor in the development of EAH by potentiating the water retention effects of AVP at the kidney (86).
At least two strategies can be used to present EAH: avoid overdrinking (real time sensation of thirst) and limiting the availability of fluids during events. Supplementation with sodium may delay of even prevent the decline in blood sodium concentration (113,114) however drinking beyond thirst (overdrinking) will not prevent hyponatremia (115), the amount of fluid ingested is more important than the amount of sodium ingested for blood sodium concentrations (116).
EAH has a complex pathogenesis and multifactorial etiology:
(86)
An athlete should consume approximately 500 to 600 mL (17 to 20 fl oz) of water 2 to 3 hours before exercise (117). By hydrating several hours prior to the exercise, there is sufficient time for urine output to return toward normal before starting the event (30).
For normal athletic events in moderate temperatures (and altitudes), it should be enough to hydrate slowly over several hours. If the body needs water urgently it can absorb some water right through the walls of the stomach. A 1% to 2% decrease in your body weight (due to water loss) will affect performance.
The threshold for reduced performance appears to be 2% body water loss of total body mass (118,119). Dehydration equivalent to 1.5% to 2% of total body mass may decrease performance up to 15% (120). We’ve seen before that 3% to 4% losses impairs muscular strength by 2% and muscular power by 3% (35), and also reduces high-intensity endurance performance (e.g., distance running) by approximately 10% (123).
Energy drinks
Energy drinks typically contain water, carbohydrates, vitamins, minerals, with the aim of increasing energy, alertness, metabolism, and/or performance (e.g., caffeine, taurine, amino acids, glucoronolactone…) (127).
Caffeine is the most common ingredient, and is absorbed in 30 – 60 minutes (128). Caffeine  stimulates the cardiovascular system and increases epinephrine output (129,130); enhance vigilance during bouts of exhaustive exercise, and periods of sustained sleep deprivation. Energy drinks with approximately 2 mg·kgBM-1caffeine consumed 10 to 60 minutes prior to anaerobic/resistance exercise may improve upper- and lower- body total lifting volume, and improve cycling and running performance (127).
stimulates the cardiovascular system and increases epinephrine output (129,130); enhance vigilance during bouts of exhaustive exercise, and periods of sustained sleep deprivation. Energy drinks with approximately 2 mg·kgBM-1caffeine consumed 10 to 60 minutes prior to anaerobic/resistance exercise may improve upper- and lower- body total lifting volume, and improve cycling and running performance (127).
Carbohydrate feeding during exercise can improve endurance capacity and performance (131,132), through maintenance of blood glucose levels, high levels of carbohydrate oxidation (1 g of carbohydrate per minute), while sparing liver and skeletal muscle glycogen (133).
Energy drinks also have a small amount of vitamins (e.g., Vitamin B6, Vitamin B12, pantothenic acid, Vitamin C) and electrolytes (e.g., sodium, potassium, phosphorus, etc.).
Energy drinks can improve mood, reaction time, and/or markers of alertness, most likely due to the ergogenic value of caffeine and/or carbohydrate.
Caffeine can elevate metabolic rate and the rate of lipolysis. 200-500 mg of caffeine (typical of thermogenic supplements) can increase acute energy expenditure (1-24 hours) (127), chronic energy expenditure (28 days) (134), and elevate plasma free-fatty acid, glycerol levels and catecholamine secretion (127, 134). The caffeine in energy drinks ranges from 80-200 mg, and it’s not conclusive whether daily use of ED would affect long-term energy balance and body composition (127).
Individuals with metabolic syndrome and or diabetes mellitus should avoid consumption of high glycemic drinks and/or foods. More importantly, individuals with known cardiovascular disease should avoid any use of energy drinks due to the cardio stimulant effects (124).
More references here than there are chopsticks in a Chinese restaurant
1. Institute of medicine. Water. In: Dietary Reference Intakes for Water, Sodium, Chloride, Potassium and Sulfate, Washington, D.C: National Academy Press, pp. 73–185, 2005.
2. Greenleaf JE & Sargent F (1965). Voluntary dehydration in man. J Appl Physiol 20, 719–724
3. Popkin B, D’Anci K & Rosenberg I (2010). Water, hydration and health. Nutr Rev 68, 439–458.
4. Wilson MM & Morley JE (2003) Impaired cognitive function and mental performance in mild dehydration. Eur J Clin Nutr 57, S24–S29.
5. Jequier E & Constant F (2010). Water as an essential nutrient: the physiological basis of hydration. Eur J Clin Nutr 64, 115–123.
6. Thornton SN (2010). Thirst and hydration: physiology and consequences of dysfunction. Physiol Behav 100, 15–21.
7. Gomez-Pinilla F (2008). Brain foods: the effects of nutrients on brain function. Nat Rev Neurosci 9, 568–578.
8. EFSA (2011) Scientific opinion on the substantiation of health claims related to water and maintenance of normal physical and cognitive functions (ID 1102, 1294, 1331), maintenance of normal thermoregulation (ID 1208) and “basic requirement of all living things” (ID 1207) pursuant to Article 13(1) of Regulation
(EC) No. 1924/2006. EFSA J 9, 2075.
9. EFSA (2010). Scientific opinion on dietary reference values for water: EFSA panel on dietetic products, nutrition, and allergies (NDA). EFSA J 8, 1459.
10. Gandy J (2012). First findings of the United Kingdom fluid intake study. Nutr Today 47, S14–S16.
11. Gibson S, Gunn P & Maughan RJ (2012). Hydration, water intake and beverage consumption habits among adults. Nutr Bull 37, 182–192.
12. Bar-David Y, Urkin J, Bar-David Z, et al. (2009). Voluntary dehydration among elementary school children residing in a hot arid environment. J Hum Nutr Diet 22, 455–460.
13. Fadda R, Rapinett G, Grathwohl D, et al. (2012). Effects of drinking supplementary water at school on cognitive performance in children. Appetite 59, 730–737.
14. Greenleaf JE (1992). Problem: thirst, drinking behavior, and involuntary dehydration. Med Sci Sports Exerc 24, 645–656.
15. Greenleaf JE & Sargent F (1965). Voluntary dehydration in man. J Appl Physiol 20, 719–724.
16. Passe D, Horn M, Stofan J, et al. (2007). Voluntary dehydration in runners despite favorable conditions for fluid intake. Int J Sport Nutr Exerc Metab 17, 284–295.
27. Lawrence E. Armstrong (2005). Hydration assessment techniques. NutrRev 63, S40–S54.
28. Lawrence E. Armstrong. Assessing Hydration Status: The Elusive Gold Standard. J Am Coll Nutr. 2007 Oct;26(5 Suppl):575S-584S.
29. Institute of Medicine and Food and Nutrition Board. In: “Dietary Reference Intakes for Water, Potassium, Sodium, Chloride and Sulfate.” Washington, DC: National Academies Press. pp 73–185, 2004.
30. Sawka MN, Burke LM, Eichner ER, Maughan RJ, Montain SJ, Stachenfeld NS. American College of Sports Medicine position stand. Exercise and fluid replacement. American College of Sports Medicine. Med Sci Sports Exerc. 2007 Feb;39(2):377-90.
34. Armstrong LE, Soto JA, Hacker FT Jr, Casa DJ, Kavouras SA, Maresh CM: Urinary indices during dehydration, exercise, and rehydration. Int J. Sport Nutr 8:345–355, 1998.
35. Armstrong LE, Maresh CM, Castellani JW, Bergeron MF, Kenefick RW, LaGasse KE, Riebe D: Urinary Indices of Hydration Status. Int J Sport Nutr 4:265–279, 1994.
36. Greenleaf JE, Morimoto T: Mechanisms controlling fluid ingestion: thirst and drinking. In Buskirk ER, Puhl SM (eds): “Body Fluid Balance: Exercise and Sport.” Boca Raton, FL: CRC Press,1996.
37. Hubbard RW, Szlyk PC, Armstrong LE: Influence of thirst and fluid palatability on fluid ingestion during exercise. In Gisolfi CV, Lamb DR (eds): “Perspectives in Exercise Sciences and Sports Medicine. Fluid Homeostasis During Exercise.” Indianapolis: Benchmark Press Inc, 1990.
38. Begum MN & Johnson CS (2010) A review of the literature on dehydration in institutionalized elderly. e-Spen Eur e-J Clin Nutr Metab 5, e47–e53.
39. Mentes J (2006). Oral hydration in older adults: greater awareness is needed in preventing, recognizing, and treating dehydration. Am J Nurs 106, 40–49.
40. Rolls BJ & Phillips PA (2009). Aging and disturbances of thirst and fluid balance. Nutr Rev 48, 137–144.
41. D’Anci K, Constant F & Rosenberg I (2006). Hydration and cognitive function in children. Nutr Rev 64, 457–464.
42. Kenney WL & Chiu P (2001). Influence of age on thirst and fluid intake. Med Sci Sports Exerc 33, 1524–1532
43. Mentes J (2006). Oral hydration in older adults: greater awareness is needed in preventing, recognizing, and treating dehydration. Am J Nurs 106, 40–49.
44. Wilson MM & Morley JE (2003) Impaired cognitive function and mental performance in mild dehydration. Eur J Clin Nutr 57, S24–S29.
45. Bourque CW (2008) Central mechanisms of osmosensation and systemic osmoregulation. Nat Rev Neurosci 9, 519–531.
46. Daniel A. Judelson , Carl M. Maresh , Linda M. Yamamoto , Mark J. Farrell , Lawrence E. Armstrong , William J. Kraemer , Jeff S. Volek , Barry A. Spiering , Douglas J. Casa , Jeffrey M. Anderson. Effect of hydration state on resistance exercise-induced endocrine markers of anabolism, catabolism, and metabolism. Journal of Applied Physiology Published 1 September 2008Vol. 105no. 3, 816-824
47. Schliess F, Ha¨ussinger D. Cell hydration and insulin signaling. Cell Physiol Biochem 10: 403–408, 2000.
48. Schliess F, Ha¨ussinger D. Cell volume and insulin signaling. Int Rev Cytol 225: 187–228, 2003.
49. Schliess F, von Dahl S, Ha¨ussinger D. Insulin resistance induced by loop diuretics and hyperosmolarity in perfused rat liver. Biol Chem 382:1063–1069, 2001.
50. Maughan R, Shirreffs S & Watson P (2007) Exercise, heat, hydration and the brain. J Am Coll Nutr 26, 604S–612S.
51. Di S & Tasker J (2004) Dehydration-induced synaptic plasticity in magnocellular neurons of the hypothalamus supraoptic nucleus. Endocrinology 145, 5141–5149.
52. Kempton MJ, Ettinger U, Foster R, et al. (2011) Dehydration affects brain structure and function in healthy adolescents.Hum Brain Mapp 32, 71–79.
55. Almond, Christopher; Shin, Andrew (2005). Hyponatremia among runners in the Boston Marathon. N Engl J Med 352 (15): 1550
56. Babar, S. (October 2013). “SIADH Associated With Ciprofloxacin.”. The Annals of Pharmacotherapy 47 (10): 1359–1363.
69. Mitchell JB, Dugas JP, McFarlin BK, Nelson MJ. Effect of exercise, heat stress, and hydration on immune cell number and function. Med Sci Sports Exerc 34: 1941–1950, 2002.
70. Powers SK, Howley ET, Cox R. A differential catecholamine response during prolonged exercise and passive heating. Med Sci Sports Exerc 14: 435–439, 1982.
71. Roy BD, Green HJ, Burnett M. Prolonged exercise following diuretic induced hypohydration effects on fluid and electrolyte hormones. Horm Metab Res 33: 540–547, 2001.
72. Roy BD, Green HJ, Burnett ME. Prolonged exercise following diuretic induced hypohydration: effects on cardiovascular and thermal strain. Can J Physiol Pharmacol 78: 541–547, 2000.
74. Tee JC, Bosch AN, Lambert MI. Metabolic consequences of exercise induced muscle damage. Sports Med 37: 827–836, 2007.
75. Del Aguila LF, Krishnan RK, Ulbrecht JS, Farrell PA, Correll PH, Lang CH, Zierath JR, Kirwan JP. Muscle damage impairs insulin stimulation of IRS-1, PI 3-kinase, and Akt-kinase in human skeletal muscle. Am J Physiol Endocrinol Metab 279: E206–E212, 2000
76. Kirwan JP, Del Aguila LF. Insulin signalling, exercise and cellular integrity. Biochem Soc Trans 31: 1281–1285, 2003.
77. Kirwan JP, Hickner RC, Yarasheski KE, Kohrt WM, Wiethop BV, Holloszy JO. Eccentric exercise induces transient insulin resistance in healthy individuals. J Appl Physiol 72: 2197–2202, 1992.
78. Asp S, Daugaard JR, Kristiansen S, Kiens B, and Richter EA. Eccentric exercise decreases maximal insulin action in humans:muscle and systemic effects. J Physiol (Lond) 494: 891–898, 1996.
79. Asp S, Kristiansen S, and Richter EA. Eccentric muscle damage transiently decreases rat skeletal muscle GLUT-4 protein. J Appl Physiol 79: 1338–1345, 1995.
80. Kirwan JP, Hickner RC, Yarasheski KE, Kohrt WM, Wiethop BV, and Holloszy JO. Eccentric exercise induces transient insulin resistance
81. Kirwan JP, Bourey RE, Kohrt WM, Staten MA, and Holloszy JO. Effects of treadmill exercise to exhaustion on the insulin response to hyperglycemia in untrained men. J Appl Physiol 70: 246–250, 1991.
82. Krishnan RK, Hernandez JM, Williamson DL, O’Gorman DJ, Evans WJ, and Kirwan JP. Age-related differences in the pancreatic b-cell response to hyperglycemia after eccentric exercise. Am J Physiol Endocrinol Metab 275: E463–E470, 1998.
83. J,Estevez E,Baquero E,Mora-Rodriguez R (2008). “Anaerobic performance when rehydrating with water or commercially available sports drinks during prolonged exercise in the heat”. Applied Physiology, Nutrition and Metabolism 33 (2): 290–298.
84. Rosner MH. Exercise-associated hyponatremia. Semin Nephrol . 2009;29:271–281.
85. Rosner MH, Bennett B, Hew-Butler T, Hoffman MD. Exercise induced hyponatremia. In: Simon EE, ed. Hyponatremia: Evaluation and Treatment . New York, NY: Springer; 2013
86. Bennett, BL; Hew-Butler, T; Hoffman, MD; Rogers, IR; Rosner, MH (Sep 2013). “Wilderness Medical Society practice guidelines for treatment of exercise-associated hyponatremia.”. Wilderness & environmental medicine 24 (3): 228–40.
87. Hew-Butler T, Ayus JC, Kipps C, et al. Statement of the Second International Exercise-Associated Hyponatremia Consensus Development Conference, New Zealand, 2007. Clin J Sport Med. 2008;18:111–121.
88. Hew TD, Chorley JN, Cianca JC, Divine JG. The incidence, risk factors, and clinical manifestations of hyponatremia in marathon runners. Clin J Sport Med . 2003;13:41–47.
89. Davis DP, Videen JS, Marino A, et al. Exercise-associated hyponatremia in marathon runners: a two-year experience.J Emerg Med. 2001;21:47–57.
90. Speedy DB, Noakes TD, Rogers IR, et al. Hyponatremia in ultra-distance triathletes. Med Sci Sports Exerc. 1999;31:809–815.
91. Lee JK, Nio AQ, Ang WH, et al. First reported cases of exercise-associated hyponatremia in Asia. Int J Sports Med . 2011;32:297–302.
92. Glace B, Murphy C. Severe hyponatremia develops in a runner following a half-marathon. JAAPA . 2008;21:27–29
93. Shapiro SA, Ejaz AA, Osborne MD, Taylor WC. Moderate exercise-induced hyponatremia.Clin J Sport Med . 2006;16:72–73
100. Speedy DB, Noakes TD, Boswell T, Thompson JM, Rehrer N, Boswell DR. Response to a fluid load in athletes with a history of exercise induced hyponatremia. Med Sci Sports Exerc. 2001;33:1434 –1442.
101. Noakes TD, Wilson G, Gray DA, Lambert MI, Dennis SC. Peak rates of diuresis in healthy humans during oral fluid overload. S Afr Med J. 2001;91:852– 857.
102. Rosner MH, Kirven J. Exercise-associated hyponatremia. Clin J Am Soc Nephrol. 2007;2:151–161.
103. Rosner MH. Exercise-associated hyponatremia. Semin Nephrol. 2009;29:271–281.
104. Hew-Butler T, Jordaan E, Stuempfle KJ, et al. Osmotic and nonosmotic regulation of arginine vasopressin during prolonged endurance exercise. J Clin Endocrinol Metab. 2008;93:2072–2078.
105. Noakes TD, Sharwood K, Speedy D, et al. Three independent biological mechanisms cause exercise-associated hyponatremia: evidence from 2,135 weighed competitive athletic performances. Proc Natl Acad Sci U S A. 2005; 102:18550 –18555.
106. Edelman IS, James AH, Brooks L, Moore FD. Body sodium and potassium. IV. The normal total exchangeable sodium; its measurement and magnitude. Metabolism. 1954;3:530 –538.
107. Edelman IS, James AH, Baden H, Moore FD. Electrolyte composition of bone and the penetration of radio sodium and deuterium oxide into dog and human bone. J Clin Invest. 1954;33:122–131.
108. Rose BD, Post TW. Clinical Physiology of Acid-Base and Electrolyte Disorders. 5th ed. New York, NY: McGraw Hill; 2001.
109. Siegel AJ, Verbalis JG, Clement S, et al. Hyponatremia in marathon runners due to inappropriate arginine vasopressin secretion. Am J Med. 2007;120:461.e11-e17.
110. Ayus JC, Varon J, Arieff AI. Hyponatremia, cerebral edema, and noncardiogenic pulmonary edema in marathon runners. Ann Intern Med. 2000;132:711–714.
111. Halperin ML, Kamel KS, Sterns R. Hyponatremia in marathon runners. N Engl J Med. 2005;353:427– 428.
112. Lindinger MI, Heigenhauser GJ, McKelvie RS, Jones NL. Blood ion regulation during repeated maximal exercise and recovery in humans. Am J Physiol. 1992;262(1 Pt 2):R126–R136.
113. Barr SI, Costill DL, Fink WJ. Fluid replacement during prolonged exercise: effects of water, saline, or no fluid. Med Sci Sports Exerc. 1991;23:811– 817.
114. Vrijens DM, Rehrer NJ. Sodium-free fluid ingestion decreases plasma sodium during exercise in the heat. J Appl Physiol. 1999;86:1847–1851.
115. Hew-Butler TD, Sharwood K, Collins M, Speedy D, Noakes T. Sodium supplementation is not required to maintain serum sodium concentrations during an Ironman triathlon. Br J Sports Med. 2006;40:255–259.
116. Weschler LB. Exercise-associated hyponatraemia: a mathematical review. Sports Med. 2005;35:899 –922
117. Casa, Douglas J., Armstrong, Lawrence E., Hillman, Susan K., Montain, Scott J. (2000). National Athletic Trainers’ Association Position Statement: Fluid replacement for athletes. Journal of Athletic Training, 35(2), 212-224.
118. Baker LB, Dougherty KA, Chow M, et al. Progressive dehydration causes a progressive decline in basketball skill performance. Med Sci Sports Exerc. 2007;39:1114–1123.
119. Hoffman JR, Stavsky H, Falk B. The effect of water restriction on anaerobic power and vertical jumping height in basketball players. Int J Sports Med. 1995;16:214–218.
120. Edwards AM, Mann ME, Marfell-Jones MJ, et al. Influence of moderate dehydration on soccer performance: physiological responses to 45 min of outdoor match-play and the immediate subsequent performance of sport-specific and mental concentration tests. Br J Sports Med. 2007;41:385–391.
121. Binkley HM, Beckett J, Casa DJ, et al. National Athletic Trainers’ Association position statement: exertional heat illnesses. J Athl Train. 2002;37:329–343.
122. Sawka M, Burke LM, Eicher ER, et al. Exercise and fluid replacement. Med Sci Sports Exerc. 39:377–390.
123. Judelson DA, Maresh CM, Anderson JM, et al. Hydration and muscular performance. Does fluid balance affect strength, power and high-intensity endurance? Sports Med. (New Zealand). 2007;37:907–921.
124. Convertino V, Armstrong LE, Coyle EF, et al. ACSM position stand: exercise and fluid replacement. Med Sci Sports Exerc. 1996;28:i–ix.
125. Coyle EF. Fluid and fuel intake during exercise. J Sports Sci. 2004;22:39–55.
126. Maughn R, Burke LM, Coyle EF, eds. Food, Nutrition and Sports Performance II. The International Olympic Committee Consensus on Sports Nutrition. London: Routledge, 2004.
127. Bill Campbell, Colin Wilborn, Paul La Bounty, Lem Taylor, Mike T Nelson, Mike Greenwood, Tim N Ziegenfuss, Hector L Lopez, Jay R Hoffman, Jeffrey R Stout, Stephen Schmitz, Rick Collins, Doug S Kalman, Jose Antonio and Richard B Kreider. International Society of Sports Nutrition position stand: energy drinks. Journal of the International Society of Sports Nutrition 2013, 10:1
128. Goldstein ER, Ziegenfuss T, Kalman D, Kreider R, Campbell B, Wilborn C, Taylor L, Willoughby D, Stout J, Graves BS, et al: International society of sports nutrition position stand: caffeine and performance. J Int Soc Sports Nutr 2010, 7:5.
129. Graham TE, Hibbert E, Sathasivam P: Metabolic and exercise endurance effects of coffee and caffeine ingestion. J Appl Physiol 1998, 85:883–889.
130. McLellan TM, Bell DG: The impact of prior coffee consumption on the subsequent ergogenic effect of anhydrous caffeine. Int J Sport Nutr Exerc Metab 2004, 14:698–708.
131. Jeukendrup A, Brouns F, Wagenmakers AJ, Saris WH: Carbohydrate-electrolyte feedings improve 1 h time trial cycling performance. Int J Sports Med 1997, 18:125–129.
132. Jeukendrup AE: Carbohydrate intake during exercise and performance. Nutrition 2004, 20:669–677.
133. Coyle EF, Coggan AR, Hemmert MK, Ivy JL: Muscle glycogen utilization during prolonged strenuous exercise when fed carbohydrate. J Appl Physiol 1986, 61:165–172.
134. Roberts MD, Dalbo VJ, Hassell SE, Stout JR, Kerksick CM: Efficacy and safety of a popular thermogenic drink after 28 days of ingestion. J Int Soc Sports Nutr 2008, 5:19.




